The World Health Organisation (WHO) has issued an urgent call for manufacturers of mpox vaccines to submit an Expression of Interest for Emergency Use Listing (EUL).
The global health body disclosed this in a statement on its website on Friday.
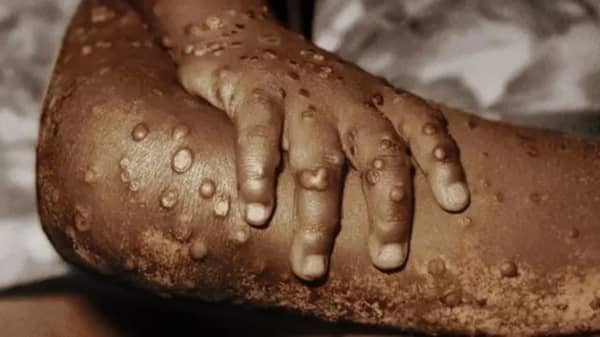
This call comes amid a growing and serious mpox outbreak in the Democratic Republic of the Congo (DRC) that has now spread beyond the country’s borders. A new viral strain, first detected in September 2023, has been identified outside the DRC for the first time.
WHO Director-General, Tedros Ghebreyesus, announced on August 7, 2024, that he had initiated the process for the EUL of mpox vaccines due to these worrying trends.
The EUL procedure is a streamlined authorisation process designed to expedite the availability of unlicensed medical products, like vaccines, during public health emergencies.
WHO is asking vaccine manufacturers to provide data demonstrating the safety, efficacy, quality, and suitability of their products for targeted populations.
“Granting an EUL will accelerate vaccine access, particularly for lower-income countries without national regulatory approval. The EUL also enables partners, including Gavi and UNICEF, to procure vaccines for distribution,” WHO said.
Mpox is a viral illness caused by the monkeypox virus. It spreads through physical contact with an infected person, contaminated materials, or infected animals.
Two vaccines have been recommended for use by the WHO Strategic Advisory Group of Experts on Immunisation (SAGE).